- On this page: Types of AMD
- Causes and Associations
- Testing
- Treatment
- Genetics
- Research
Age-related macular degeneration (AMD) is a common condition among people ages 50 years and older and affects approximately 11 million people in the United States. The incidence of AMD increases with each decade over age 50.
AMD affects the macula, the central portion of the retina, which is the light-sensing nerve tissue that lines the inside of the back of the eye. The retina, along with the optic nerve and brain, transforms and processes light into visual images.
Many people with AMD have minimal visual symptoms and may retain good vision. A small percentage of those with AMD will lose central vision, which may impair their ability to perform activities such as reading and driving.
The two major types of AMD are dry and wet.
Dry (atrophic) AMD affects about 90 percent of all people with AMD.
In dry AMD, the macula becomes thinner and develops drusen, which are small yellow deposits under the retina. Most people with drusen alone do not have significant visual changes or vision loss.
There are three stages of dry AMD: early, intermediate, and late. Dry AMD can turn into wet AMD at any stage. A minority of people with dry AMD will develop the late stage, characterized by geographic atrophy.
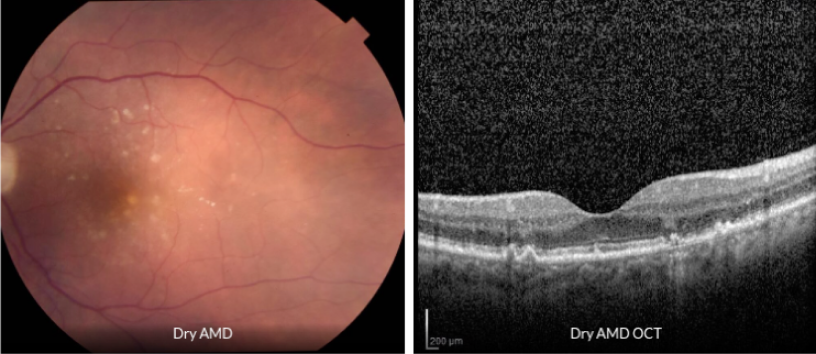
Many people with mild dry AMD have little to no visual symptoms; however, significant loss of central vision can occur in later stages. Peripheral vision is typically not affected. Therefore, complete blindness from AMD alone is very unlikely.
People with dry AMD may experience:
A minority of people with dry AMD will advance to the late stage, which is characterized by geographic atrophy and can result in central vision loss. Geographic atrophy involves the loss of pigmented cells beneath the macula. These pigmented cells normally act to support and nourish the photoreceptor cells of the retina, which produce color and low-light vision. Without these pigmented support cells, the photoreceptors no longer function, leading to blind spots in the central vision.
Wet (neovascular) AMD affects only about 10 percent of people with AMD, yet accounts for the majority of AMD-related central vision loss.
Wet AMD is characterized by choroidal neovascularization (CNV), which is the abnormal growth of blood vessels that can cause bleeding or swelling in the macula. If left untreated, CNV typically results in permanent scarring of macular tissue and severe central vision loss.
While there is no cure for wet AMD, there are treatments that are very effective and can improve or stabilize vision.
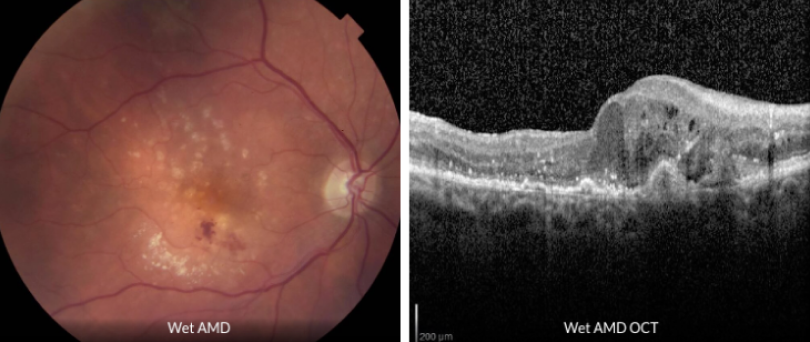
Those with wet AMD often have rapid progressive loss of central vision, typically over days to weeks.
People with wet AMD may experience:
There are no known specific causes of AMD, though there does appear to be a genetic component in many cases. It is a degenerative condition that occurs over time and is typically found in people ages 50 years or older.
The following healthy choices can prevent or slow the progression of macular degeneration:
A comprehensive eye examination is important to assess AMD, including vision testing, eye drops to dilate the pupils, and a complete examination of the front and back of the eye. A driver should accompany the patient during their eye exam as pupillary dilation may create near-vision blurring.
People with AMD may undergo several types of tests to assess the disease, such as:
While dry AMD is not curable, treatment and preventative measures can help reduce the risk of advanced disease. Intravitreal injections can slow the progression of geographic atrophy that appears in the late stage of dry AMD.
Intravitreal Injections
Intravitreal injections are available to treat late-stage dry AMD when geographic atrophy is present. With this treatment, Syfovre, an inhibitor of the complement pathway involved in inflammation, is injected directly into the eye.
This procedure is performed in the office and requires only a local anesthetic. Intravitreal injections may be administered every one to two months and may help slow down the growth rate of the geographic atrophy to delay further vision loss. It is important to understand that this treatment will not improve or restore vision.
Vitamins
Studies have demonstrated that AREDS and AREDS2 (antioxidant vitamins and minerals) formulas can reduce the risk of progression of dry AMD to wet AMD. Smokers should avoid the original AREDS formula containing beta-carotene, which has been associated with an increased risk of lung cancer in those who smoke. The AREDS 2 formula is safe regardless of an individual’s smoking history. An eye care provider can help patients determine if vitamin supplementation is needed.
While wet AMD is not fully curable, treatment can preserve vision and keep the condition from progressing. Treatments include medications aimed at blocking growth factors, nondestructive laser-drug combinations, and traditional laser photocoagulation.
Intravitreal Injections
Intravitreal injections are commonly used to treat AMD. With this treatment, anti-vascular endothelial growth factor (VEGF) inhibitors such as Lucentis, Eylea, Avastin, Beovu, and Vabysmo are injected directly into the eye.
This procedure is performed in the office and requires only a local anesthetic. Intravitreal injections may be administered as frequently as once a month, depending on the nature of the disease, in order to maintain eye health and optimize vision.
Some patients may see an improvement in their vision from these injections as well, depending on their disease progression.
Photodynamic Therapy
Photodynamic therapy (PDT) is an office-based procedure that utilizes an intravenous injection of a photosensitizing medication and a laser light to reduce leakage from choroidal neovascularization. Unlike anti-VEGF therapy, PDT does not improve vision and is now seldom used except in rare cases.
Laser Photocoagulation
Laser photocoagulation is an office-based procedure that uses heat to destroy choroidal neovascularization. Unlike anti-VEGF therapy, laser photocoagulation does not improve vision and is now seldom used except in rare cases.
Genetics plays a key role in AMD. Those with a family history of AMD have a 70 percent risk of developing the disease.
Researchers have recently identified genetic markers that strongly influence AMD disease progression. These gene variants can manipulate the immune system, metabolic enzymes, or mitochondrial function to accelerate AMD progression through micro-cellular inflammation and increased oxidative stress in the retina. Many of these discoveries are leading to new therapies to slow down the progression of AMD.
Early identification and frequent monitoring of individuals with these genetic markers may result in early detection of AMD, leading to better visual outcomes through earlier treatment.
As a center of excellence for macular degeneration, Mid Atlantic Retina physicians are closely involved in the latest clinical trials and new therapeutic approaches for AMD.
To learn more about our AMD clinical trials, visit our Research page.
An Amsler grid is a self-monitoring tool that allows patients with dry AMD to check for visual blurring or distortion that may signify the transition from dry to wet AMD. Patients with visual changes on the Amsler grid should contact their ophthalmologist or retina specialist promptly.
Click here to download the Amsler Grid Chart.
If you have any signs or symptoms of macular degeneration, contact us today to meet with a Mid Atlantic Retina specialist. While there is currently no cure for AMD, our team can work with you to protect your vision and minimize your complications.
Macular degeneration vision loss is the most common cause of vision loss in adults over...
Macular degeneration is the most common cause of vision loss in individuals aged 60 and...